 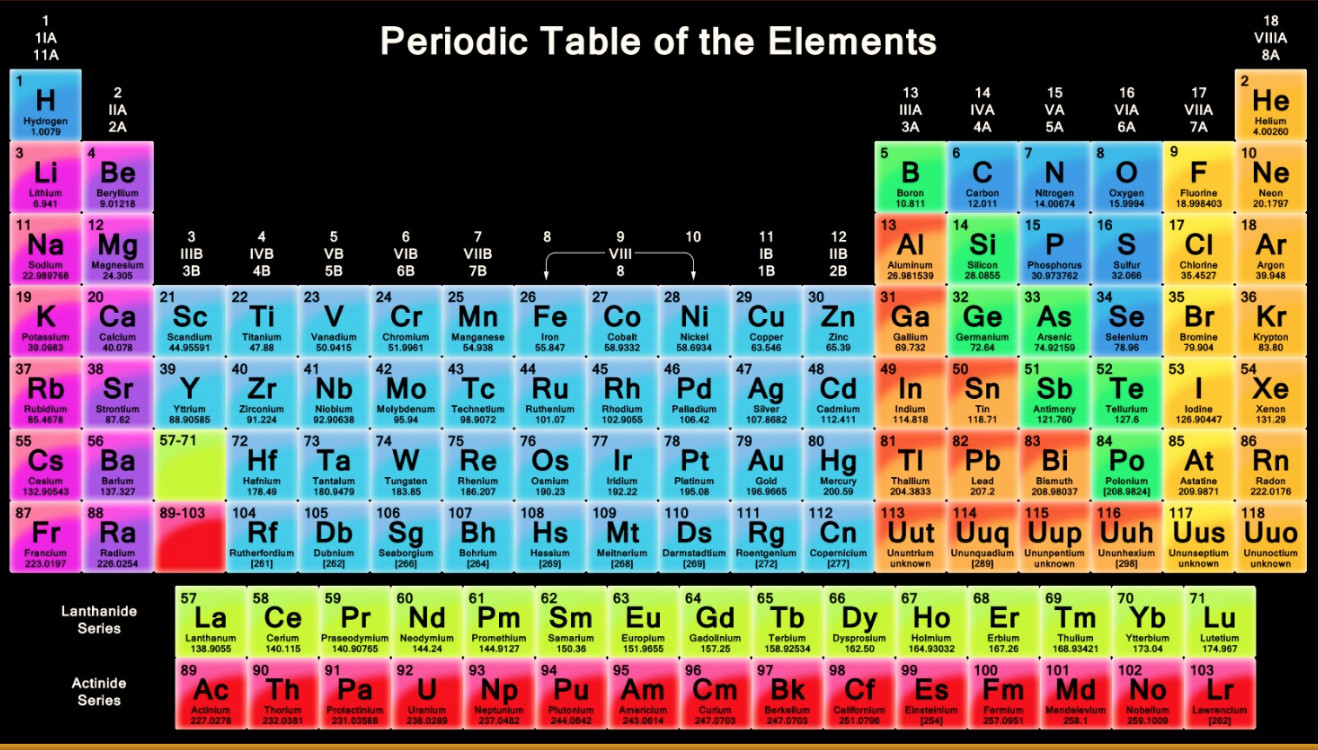
The system of eighteen groups is generally accepted by the chemistry community, but some dissent exists about membership of elements number 1 and 2 ( hydrogen and helium). 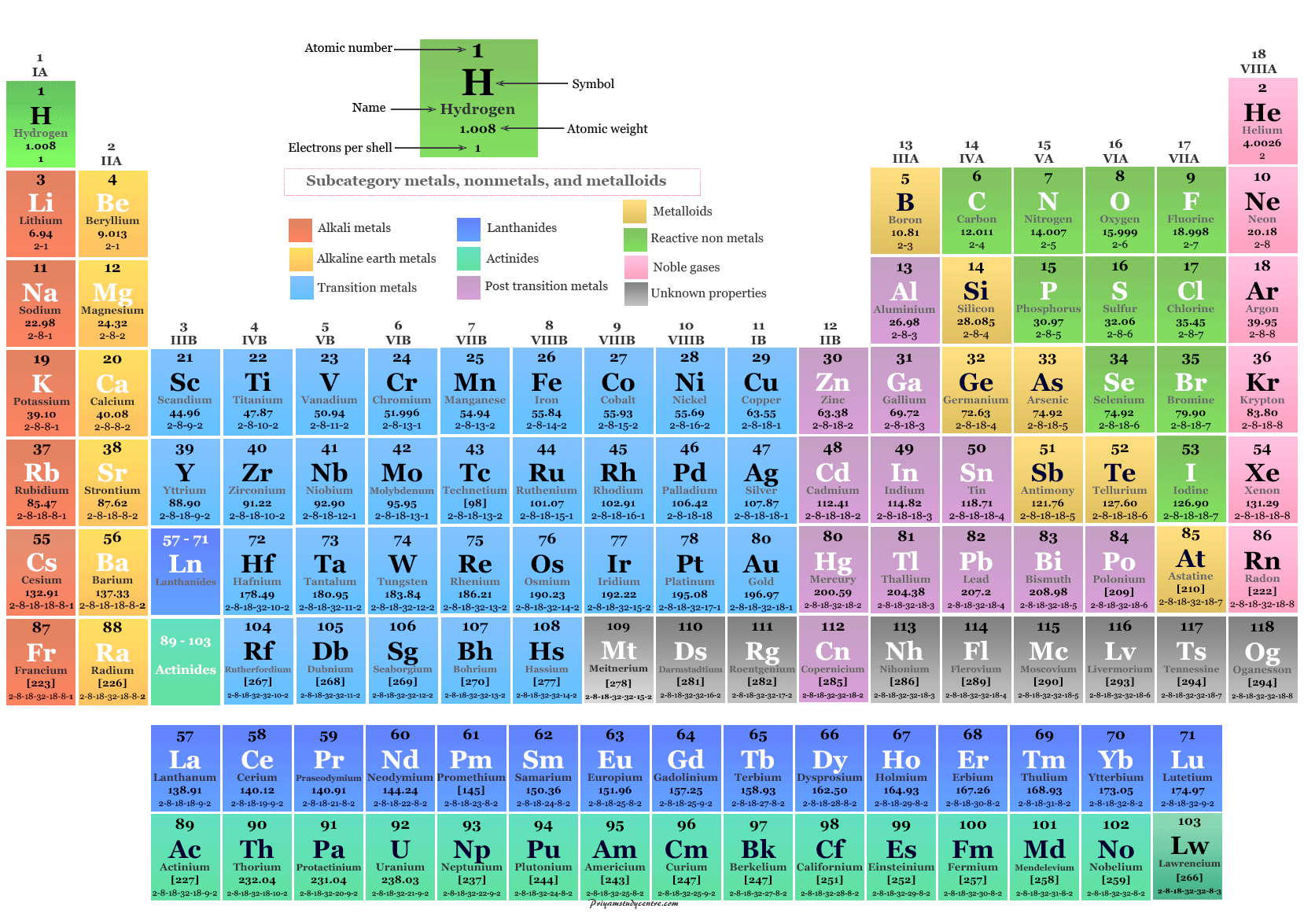
It replaces two older incompatible naming schemes, used by the Chemical Abstract Service (CAS, more popular in the United States), and by IUPAC before 1988 (more popular in Europe). The modern numbering system of "group 1" to "group 18" has been recommended by the International Union of Pure and Applied Chemistry (IUPAC) since 1988. There are three systems of group numbering for the groups the same number may be assigned to different groups depending on the system being used. The elements in a group have similar physical or chemical characteristics of the outermost electron shells of their atoms (i.e., the same core charge), because most chemical properties are dominated by the orbital location of the outermost electron. There are 18 numbered groups in the periodic table the 14 f-block columns, between groups 2 and 3, are not numbered. In chemistry, a group (also known as a family) is a column of elements in the periodic table of the chemical elements. In the periodic table of the elements, each column is a group. Figure 4: The ionic radius increases for the generation of negative ions.Column of elements in the periodic table of the chemical elements The gain of an electron adds more electrons to the outermost shell which increases the radius because there are now more electrons further away from the nucleus and there are more electrons to pull towards the nucleus so the pull becomes slightly weaker than of the neutral atom and causes an increase in atomic radius. Figure 3: The ionic radius decreases for the generation of positive ions.Īn anion, on the other hand, will be bigger in size than that of the atom it was made from because of a gain of an electron. A neutral atom X is shown here to have a bond length of 180 pm and then the cation X + is smaller with a bond length of 100 pm. This can similarly be said about the protons pulling the electrons closer to the nucleus, which as a result decreases atomic size.įigure 3 below depicts this process. If ten magnets and ten metallic objects represent a neutral atom where the magnets are protons and the metallic objects are electrons, then removing one metallic object, which is like removing an electron, will cause the magnet to pull the metallic objects closer because of a decrease in number of the metallic objects. An analogy to this can be of a magnet and a metallic object.It will also decrease because there are now less electrons in the outer shell, which will decrease the radius size. 
This will cause a decrease in atomic size because there are now fewer electrons for the protons to pull towards the nucleus and will result in a stronger pull of the electrons towards the nucleus.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
